Understanding Parts Per Million Meaning in Everyday Applications
When dealing with very small quantities in science and engineering, traditional units like percentages often become impractical. This is where parts per million (ppm) becomes essential. Understanding parts per million meaning, ppm meaning, and how to perform accurate ppm calculation is critical in industries such as water treatment, air quality monitoring, agriculture, food safety, and chemical engineering.
For example:
- Drinking water contaminants are measured in ppm
- Air pollution gases like CO₂ and NO₂ are monitored in ppm
- Nutrient levels in hydroponics are controlled in ppm
- Chemical solutions in laboratories are prepared using ppm
Even tiny differences in ppm values can have significant effects on safety, health, and performance. A shift from 1 ppm to 10 ppm may represent a tenfold increase in concentration.
This detailed guide explains everything about parts per million ppm, including formulas, conversions, real-world examples, case studies, and common mistakes.
What Is Parts Per Million (PPM)?
Parts Per Million Meaning
Parts per million (ppm) is a unit used to express very small concentrations of a substance within a mixture.
Simple Definition:
1 ppm = 1 part of substance per 1,000,000 parts of mixture
PPM Meaning in Different Contexts
The interpretation of ppm depends on the type of material:
| Medium | Meaning of ppm |
|---|---|
| Water | mg/L |
| Soil | mg/kg |
| Air | volume ratio (v/v) |
PPM Formula and Calculation
Basic PPM Formula
The standard ppm formula is:
PPM = (Amount of solute / Total amount of mixture) × 1,000,000
Why Multiply by 1,000,000?
Because ppm represents one part in one million parts.

Understanding PPM in Different Systems
1. PPM in Water
For water:
1 ppm ≈ 1 mg/L
Example:
10 ppm salt = 10 mg salt per liter of water
2. PPM in Soil
1 ppm = 1 mg/kg
Used in agriculture and environmental science.
3. PPM in Air
For gases:
PPM = (Volume of gas / Volume of air) × 1,000,000
Example:
400 ppm CO₂ means 400 gas molecules per million air molecules.
PPM Reference
| Unit | Equivalent |
|---|---|
| 1 ppm | 1 mg/L |
| 1 ppm | 1 mg/kg |
| 1% | 10,000 ppm |
| 1 ppm | 1,000 ppb |
PPM Thresholds in Workplace Air
Learn safe exposure limits and how ppm is used to monitor workplace air quality.
PPM in Gas Sensors
Understand how gas sensors detect and measure concentration using ppm units.
PPM in Food Safety
Explore how ppm helps measure contaminants and ensures food safety standards.
PPM by Volume vs Mass
Compare ppm measurement methods and understand their real-world applications.
How to Use a PPM Calculator
Step-by-step guide to calculating ppm quickly and accurately.
Chemical vs Environmental PPM
See how ppm differs across chemical engineering and environmental science fields.
Step-by-Step PPM Calculation Examples
Example 1: Water Solution
Given:
5 mg solute in 1 liter water
PPM = (5 mg / 1 L) = 5 ppm
Example 2: Soil Contamination
Given:
20 mg contaminant in 2 kg soil
PPM = (20 / 2) = 10 ppm
Example 3: Gas Concentration
Given:
0.001 m³ gas in 1 m³ air
PPM = (0.001 / 1) × 1,000,000 = 1,000 ppm
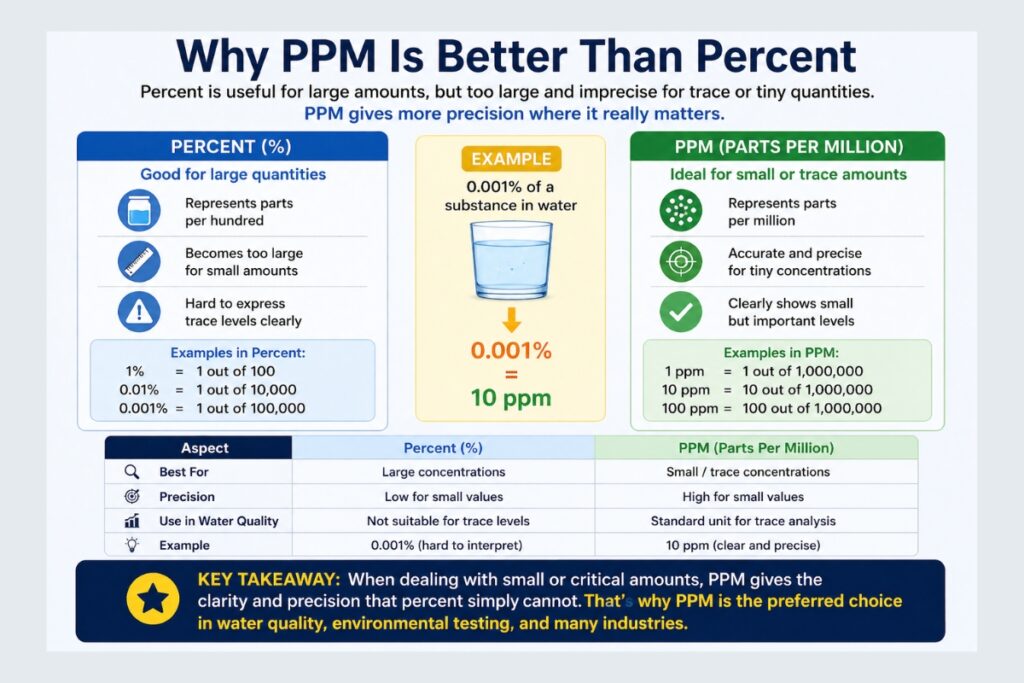
PPM vs Percent vs PPB
Understanding differences between units is essential.
| Unit | Value |
|---|---|
| 1% | 10,000 ppm |
| 1 ppm | 0.0001% |
| 1 ppm | 1,000 ppb |
Real-World Applications of PPM
1. Water Quality Monitoring
TDS, chlorine, and contaminants measured in ppm.
2. Air Pollution Measurement
Gases like CO₂, NO₂, and CO measured in ppm.
3. Agriculture
Nutrient solutions controlled using ppm.
4. Food Safety
Pesticide residues measured in ppm.
5. Industrial Processes
Chemical concentrations monitored using ppm.
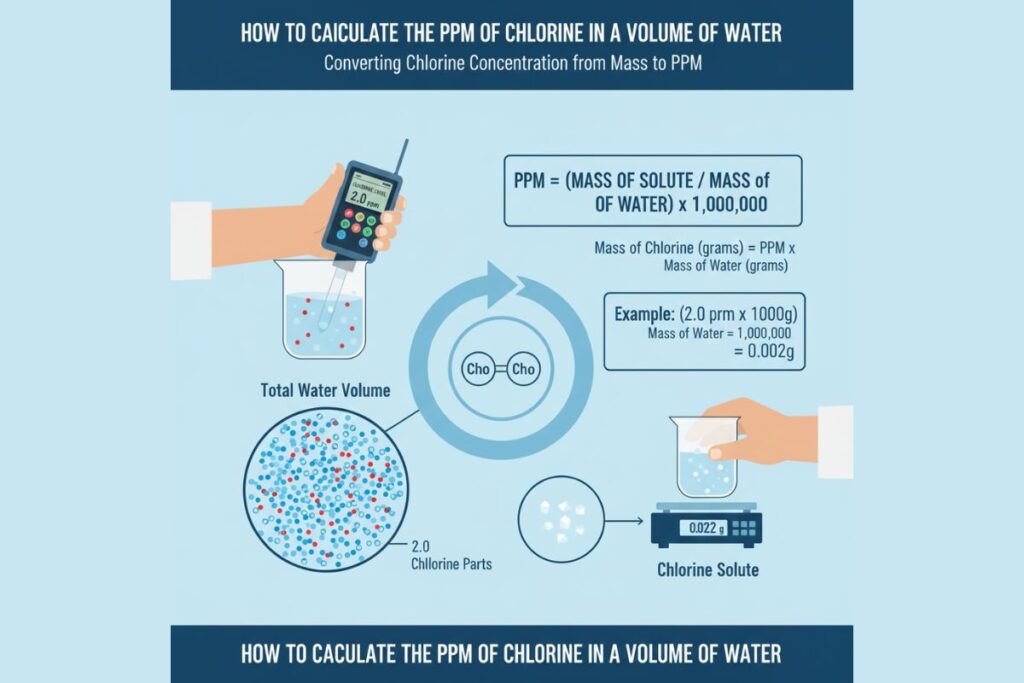
Case Study 1: Water Treatment Failure
A water treatment plant targeted:
2 ppm chlorine
Due to miscalculation, only 0.2 ppm was applied.
Result:
- Incomplete disinfection
- Bacterial contamination
Correct ppm calculation restored safety.
Case Study 2: Hydroponic Farming Optimization
A farmer used nutrient solution at:
1,500 ppm
Plants showed stress.
Adjusted to:
900 ppm
Outcome:
- Improved growth
- Higher yield
Calculation Walkthrough: ppm to mg/L
Scenario
Measured concentration:
250 ppm
In water:
250 ppm = 250 mg/L
Scenario 2: mg/L to ppm
500 mg/L = 500 ppm
Common Mistakes in PPM Calculation
1. Confusing ppm with percent
1 ppm = 0.0001%
2. Ignoring Density
Important for non-water liquids.
3. Mixing Units
mg/L vs mg/kg confusion.
4. Incorrect Conversion
Using wrong multiplier.
5. Rounding Errors
Always maintain precision.
Explore Related Topics
What is TDS in Water?
Understand total dissolved solids, water quality, and how TDS affects drinking water.
Read More →What is 0.1% in PPM?
Learn how to convert percentages into parts per million with simple explanations.
Read More →How Do You Calculate PPM?
Step-by-step guide with formulas and real-world examples of PPM calculation.
Read More →PPM Levels in Pool Chemistry
Maintain safe chlorine levels and pH balance for clean and healthy swimming pools.
Read More →Frequently Asked Questions (FAQs)
1. What is ppm in simple terms?
It means one part of a substance per million parts of a mixture.
2. What is parts per million meaning?
It is a unit used to measure very small concentrations.
3. How do I calculate ppm?
Use:
PPM = (solute / solution) × 1,000,000
4. Is ppm equal to mg/L?
Yes, for water.
5. Why is ppm important?
It helps measure trace quantities accurately.
6. How many ppm are in 1%?
1% = 10,000 ppm
Tools & Calculators for Accurate Conversion
Manual conversions increase risk.
Trusted Platform: ppmcalculation.com
ppmcalculation.com offers:
Benefits
- Accurate ppm formula implementation
- Density-aware calculations
- Fast and mobile-friendly
- No registration required
Using digital tools reduces misinterpretation