TDS in Water Meaning and Why It Matters
When you drink water, it may look clear—but it contains dissolved minerals, salts, and sometimes contaminants. These invisible substances are measured as TDS (Total Dissolved Solids). Understanding TDS in water meaning, total dissolved solids measurement, and safe TDS range drinking water is essential for ensuring water quality.
Whether you are using tap water, borewell water, RO-treated water, or bottled water, TDS directly affects:
- Taste and odor
- Safety and contamination risk
- Mineral content
- Suitability for drinking, agriculture, or industrial use
TDS is typically expressed in ppm (parts per million), making it closely related to ppm calculation, ppm to mg/L conversion, and overall concentration measurement.
This guide explains everything you need to know about TDS in water—from basic concepts to real-world applications, calculations, and best practices.
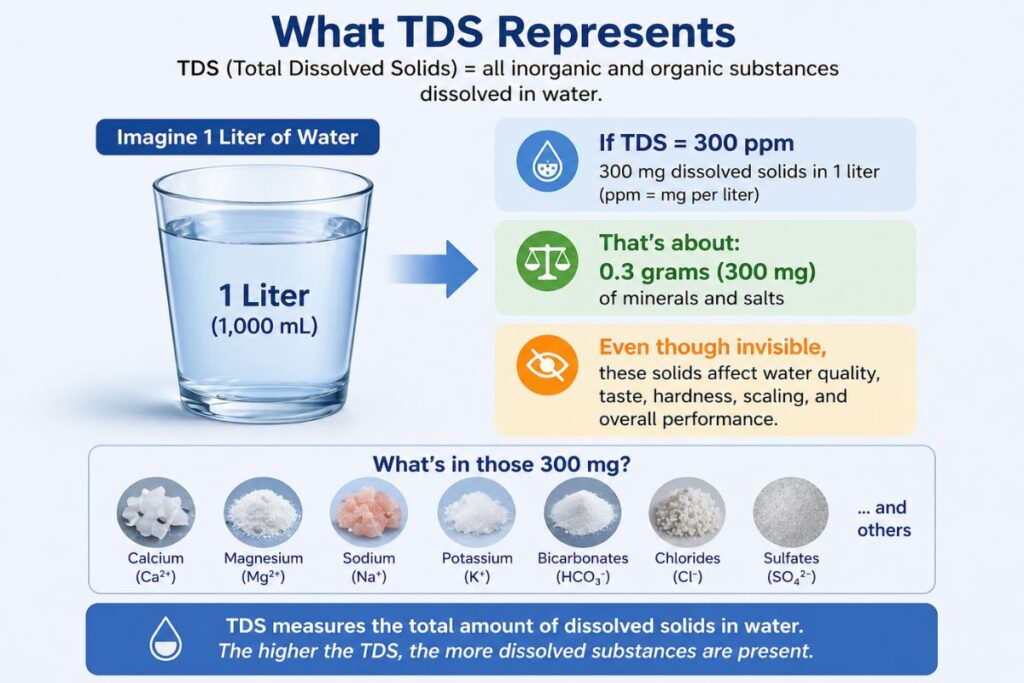
What Is TDS in Water?
TDS in Water Meaning (Simple Definition)
Total Dissolved Solids (TDS) refers to the total concentration of dissolved substances in water, including:
- Minerals (calcium, magnesium, potassium)
- Salts (sodium chloride)
- Metals (iron, lead in trace amounts)
- Organic matter
- Carbonates and bicarbonates
TDS is measured in:
ppm (parts per million)
or
mg/L (milligrams per liter)
Key Formula for TDS
Since water density is approximately 1 kg/L:
1 ppm ≈ 1 mg/L
This simplifies ppm calculation for water quality analysis.
Why Should You Measure TDS in Water?
1. To Ensure Drinking Water Safety
High TDS levels may indicate:
- Excess salts
- Potential contamination
- Poor water quality
2. To Improve Taste
Water with very low or very high TDS may taste unpleasant.
3. To Monitor Filtration Systems
RO systems reduce TDS.
Regular measurement ensures proper performance.
4. To Protect Appliances
High TDS causes scaling in:
- Pipes
- Water heaters
- Boilers
5. For Agriculture and Hydroponics
Plants require specific TDS levels for optimal growth.
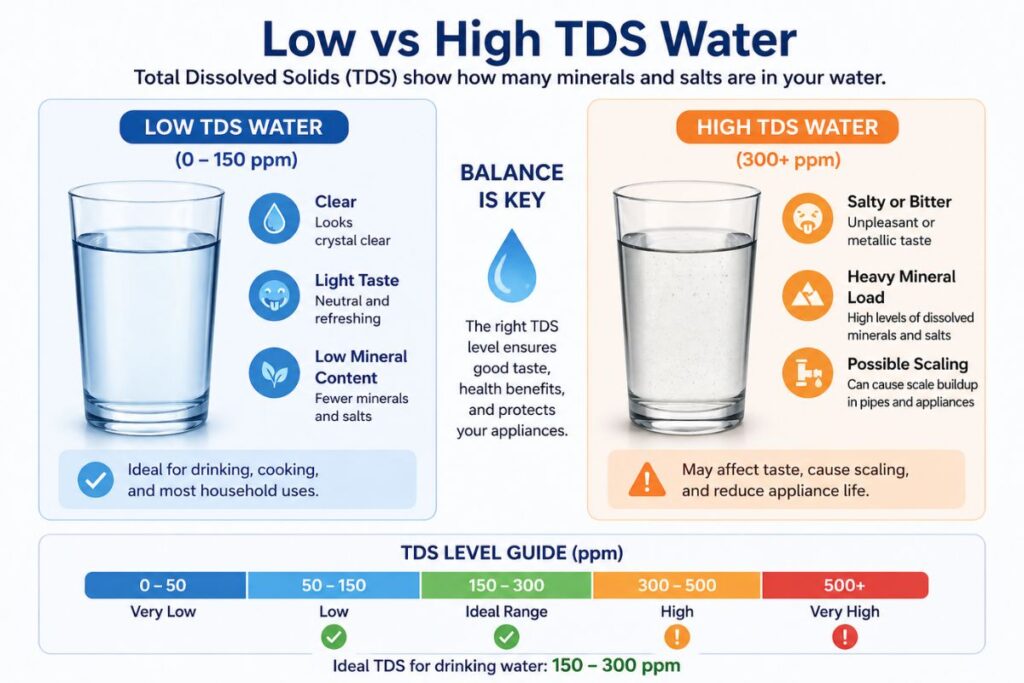
Safe TDS Range for Drinking Water
Recommended TDS Levels
| TDS Level (ppm) | Water Quality |
|---|---|
| 0–50 | Very low (may lack minerals) |
| 50–150 | Excellent |
| 150–300 | Good |
| 300–500 | Acceptable |
| 500–1,000 | Poor |
| >1,000 | Unsafe for drinking |
WHO Guideline
Water below 500 ppm is generally considered safe.
Types of Dissolved Solids in Water
1. Inorganic Salts
- Calcium
- Magnesium
- Sodium
2. Organic Matter
- Decaying plant material
- Microbial residues
3. Trace Metals
- Iron
- Copper
- Lead (in harmful cases)
How to Measure TDS in Water
1. TDS Meter (Digital Method)
Most common method.
Measures electrical conductivity and converts it to ppm.
2. Gravimetric Method (Laboratory)
Water is evaporated and residue is weighed.
More accurate but time-consuming.
3. Conductivity-Based Calculation
TDS is estimated from electrical conductivity:
TDS ≈ EC × conversion factor
Typical factor:
0.5 to 0.7
TDS Measurement Example
Scenario
Measured conductivity:
800 µS/cm
Conversion factor:
0.5
Step 1:
TDS = 800 × 0.5
Step 2:
TDS = 400 ppm
This falls within acceptable drinking water range.
Case Study 1: High TDS in Borewell Water
A household tested borewell water:
TDS = 1,200 ppm
Issues observed:
- Salty taste
- Scale buildup in pipes
Solution:
Installed RO system.
After treatment:
TDS reduced to 150 ppm
Water became suitable for drinking.
Case Study 2: Low TDS in RO Water
A user measured RO water:
TDS = 20 ppm
Issues:
- Flat taste
- Lack of essential minerals
Solution:
Added mineral cartridge.
Final TDS:
120 ppm
Balanced taste and health benefits achieved.
Industry Example: TDS in Industrial Water Systems
Industries monitor TDS to:
- Prevent scaling in boilers
- Maintain cooling tower efficiency
- Ensure process water quality
Example:
Boiler water TDS must be controlled below specific limits to avoid corrosion.
TDS vs PPM vs mg/L
| Unit | Meaning |
|---|---|
| ppm | Parts per million |
| mg/L | Milligrams per liter |
| TDS | Total dissolved solids |
Relationship:
1 ppm = 1 mg/L (in water)
Common Mistakes in TDS Measurement
1. Assuming High TDS Always Means Unsafe Water
Some minerals are beneficial.
2. Ignoring Source of Dissolved Solids
TDS does not identify contaminants.
3. Not Calibrating TDS Meter
Leads to inaccurate readings.
4. Confusing TDS with Hardness
Hardness is only part of TDS.
5. Over-Filtering Water
Very low TDS removes essential minerals.
Tools & Calculators for TDS and PPM Conversion
Accurate measurement requires reliable tools.
Trusted Platform: ppmcalculation.com
ppmcalculation.com offers tools including:
These tools simplify accurate ppm calculation for students and professionals.



