Why PPM Levels in Pool Chemistry Matter
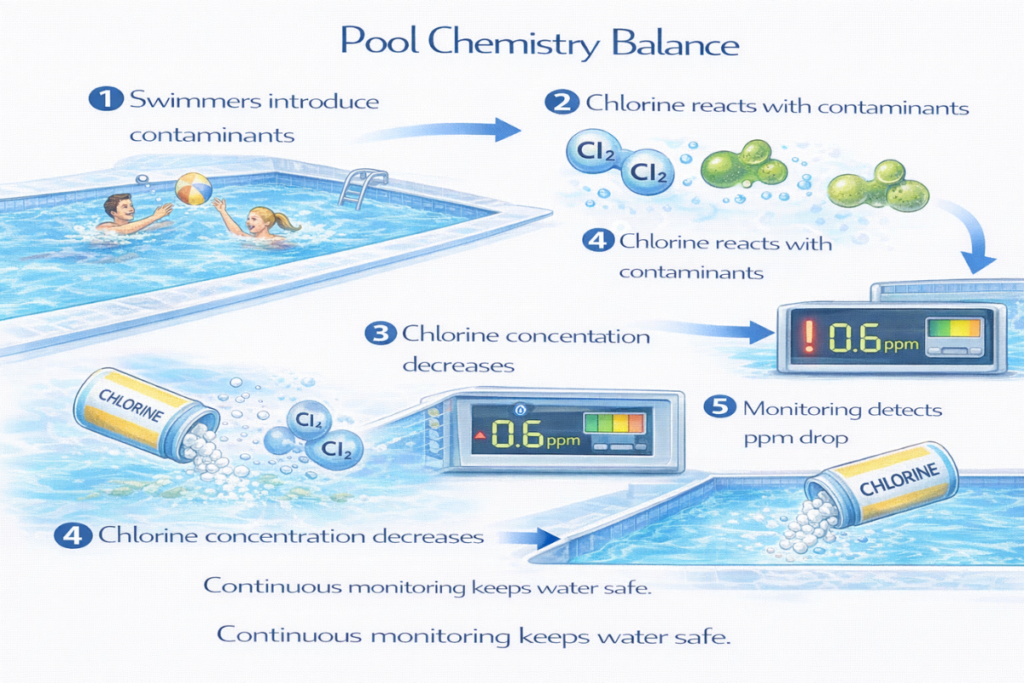
Swimming pools appear crystal clear when properly maintained, but maintaining safe water quality requires careful control of chemical concentrations. Pool operators and homeowners rely on PPM Levels in Pool Chemistry to ensure that disinfectants, stabilizers, and dissolved minerals remain within safe ranges.
If pool chemistry is not properly balanced:
- Water can become unsafe for swimmers
- Algae growth may occur
- Eye and skin irritation can increase
- Equipment corrosion may develop
This is why pool maintenance professionals measure chemicals such as chlorine, cyanuric acid, calcium hardness, and total dissolved solids in parts per million (ppm).
Even a small deviation in ppm—such as chlorine rising from 3 ppm to 10 ppm—can significantly affect swimmer comfort and safety.
Understanding ppm in pool chemistry allows pool owners to:
- Maintain effective disinfection
- Protect swimmer health
- Prevent algae growth
- Extend pool equipment lifespan
This guide explains how ppm works in pool water chemistry, ideal concentration ranges, measurement methods, and how to adjust levels correctly.
What Does PPM Mean in Pool Chemistry?
Definition of Parts Per Million in Pool Water
In swimming pool chemistry, ppm represents milligrams of a substance per liter of water.
Standard ppm formula:
PPM = (Mass of chemical / Mass of water) × 1,000,000
Because the density of water is approximately 1 kg per liter:
1 ppm ≈ 1 mg/L
This simplifies ppm calculation for pool chemicals.
Key Chemicals Measured in Pool Chemistry
Pool water contains several important chemical components.
The most commonly monitored include:
- Free chlorine
- Combined chlorine
- Cyanuric acid (stabilizer)
- Calcium hardness
- Total dissolved solids (TDS)
- Alkalinity
Each parameter is measured in ppm to ensure safe and balanced water.
Ideal PPM Ranges for Pool Water
| Parameter | Ideal Range |
|---|---|
| Free chlorine | 1–3 ppm |
| Combined chlorine | <0.5 ppm |
| Cyanuric acid | 30–50 ppm |
| Calcium hardness | 200–400 ppm |
| Total alkalinity | 80–120 ppm |
| Total dissolved solids | <1,500 ppm |
Maintaining these ranges ensures proper disinfection and water clarity.

Free Chlorine vs Combined Chlorine
Free Chlorine
Active disinfectant that kills bacteria and viruses.
Safe range:
1–3 ppm
Combined Chlorine
Occurs when chlorine reacts with contaminants such as sweat and urine.
Levels should remain below:
0.5 ppm
Higher levels cause:
- Chlorine odor
- Eye irritation
- Reduced disinfection efficiency
The Role of pH in Pool Chemistry
Although pH is not measured in ppm, it significantly influences chlorine effectiveness.
Ideal pH range:
7.2–7.6
If pH rises above 8.0:
Chlorine becomes less effective, even if ppm levels appear correct.
Calculation Walkthrough: Adding Chlorine to a Pool
Scenario
Pool size:
40,000 liters
Current chlorine level:
0.5 ppm
Target chlorine level:
2 ppm
Step 1: Calculate required increase
2 − 0.5 = 1.5 ppm
Step 2: Convert ppm to mg/L
1.5 ppm = 1.5 mg/L
Step 3: Calculate total chlorine needed
1.5 mg × 40,000 L = 60,000 mg
Step 4: Convert to grams
60 g chlorine required
This calculation ensures precise chemical dosing.
Case Study 1: Algae Growth in Residential Pool
A homeowner noticed green water despite adding chlorine.
Test results:
Free chlorine = 0.2 ppm
Recommended range:
1–3 ppm
Action taken:
Pool shock treatment increased chlorine to 4 ppm temporarily.
Result:
Algae eliminated and pool returned to safe operating levels.
Case Study 2: Chlorine Overdose in Hotel Pool
Hotel maintenance staff mistakenly added excessive chlorine.
Measured concentration:
10 ppm
Symptoms reported:
- Eye irritation
- Strong chlorine odor
Solution:
Pool diluted with fresh water until chlorine returned to 3 ppm.
This restored swimmer comfort.
Industry Example: Public Swimming Pool Management
Public pools often use automated monitoring systems.
Sensors continuously measure:
- Free chlorine ppm
- pH
- temperature
Automated dosing systems maintain ideal chemical balance.
This technology reduces human error and improves safety.
Safe vs Unsafe Pool Chemical Levels
| Parameter | Safe Level | Risk Level |
|---|---|---|
| Chlorine | 1–3 ppm | >5 ppm |
| Combined chlorine | <0.5 ppm | >1 ppm |
| Cyanuric acid | 30–50 ppm | >100 ppm |
| Calcium hardness | 200–400 ppm | >500 ppm |
Maintaining safe ppm levels prevents water quality issues.
Ideal Chlorine PPM Levels for Swimming Pools
Maintaining correct chlorine ppm helps:
✔ Kill harmful microorganisms
✔ Prevent algae formation
✔ Reduce swimmer irritation
✔ Maintain water clarity
✔ Ensure effective pool sanitation
Proper ppm management ensures both safety and comfort.
Methods for Measuring PPM in Pool Water
1. Test Strips
Quick and easy method for home users.
2. Liquid Reagent Kits
More accurate and widely used by professionals.
3. Digital Pool Testers
Electronic sensors measure concentration precisely.
These tools provide reliable concentration measurement.
Common Mistakes in Pool Chemistry
1. Ignoring Regular Testing
Chemical levels change rapidly due to sunlight and swimmers.
2. Overusing Chlorine
Excessive chlorine causes irritation and strong odor.
3. Neglecting Cyanuric Acid Levels
Low stabilizer levels allow chlorine to degrade quickly in sunlight.
4. Misinterpreting Test Results
Incorrect readings lead to improper chemical dosing.
5. Ignoring Total Dissolved Solids
High TDS reduces chemical efficiency.
Tools & Calculators for Pool Chemical Management
Precise calculations improve pool maintenance.
Trusted Platform: ppmcalculation.com
ppmcalculation.com offers tools including:
Advantages
- Accurate ppm formula implementation
- Instant calculation
- Mobile-friendly design
- No registration
- Suitable for students and professionals
Frequently Asked Questions (FAQs)
1. What is the ideal chlorine ppm for pools?
Between 1 and 3 ppm for most swimming pools.
2. Why does chlorine smell strong in some pools?
High combined chlorine levels produce chloramine odor.
3. Can high ppm chlorine harm swimmers?
Yes, excessive chlorine may cause eye and skin irritation.
4. How often should pool ppm levels be checked?
At least 2–3 times per week, especially during heavy use.
5. Does sunlight affect chlorine ppm?
Yes, UV light breaks down chlorine rapidly.
6. What happens if ppm levels are too low?
Water becomes vulnerable to bacterial contamination and algae growth.
Why PPM Levels in Pool Chemistry Are Essential
Maintaining proper PPM Levels in Pool Chemistry is the foundation of safe and hygienic swimming pool operation.
Accurate ppm monitoring helps pool owners:
✔ Maintain effective water disinfection
✔ Prevent algae and bacterial growth
✔ Protect swimmers from irritation
✔ Extend pool equipment lifespan
✔ Ensure balanced water chemistry
Understanding ppm calculation and using reliable measurement tools ensures that swimming pools remain clean, safe, and comfortable.
Take control of your pool water chemistry with accurate ppm tools.
👉 Visit ppmcalculation.com to access:
- ppm calculation tools
- ppm converter utilities
- ppm to mg/L calculators
- concentration measurement resources
Maintain perfect pool chemistry with precision using ppmcalculation.com.
Related PPM Calculators
Explore more water quality and chemistry tools:



