Why PPM in Gas Sensors Matters for Safety and Monitoring
Invisible gases can pose serious risks in homes, workplaces, and industrial facilities. Carbon monoxide leaks, methane accumulation, or poor indoor ventilation may occur without any visible signs. This is why modern monitoring systems rely on PPM in Gas Sensors to detect extremely small concentrations of gases before they become dangerous.
Gas sensors measure concentrations of substances such as:
- Carbon dioxide (CO₂)
- Carbon monoxide (CO)
- Methane (CH₄)
- Nitrogen dioxide (NO₂)
- Volatile organic compounds (VOCs)
These measurements are usually expressed in parts per million (ppm), a precise unit that allows scientists and engineers to detect trace amounts of gases in the air.
Understanding how ppm readings are generated helps users interpret sensor data correctly and improve air safety.
This guide explains how gas sensors work, how ppm values are calculated, and how industries use these technologies for environmental monitoring and safety.
What Is PPM in Gas Sensors?
Definition of PPM for Gas Measurements
Parts per million (ppm) expresses the volume of a gas relative to the total air volume.
Standard expression:
PPM = (Volume of gas / Volume of air) × 1,000,000
Because gases mix uniformly, ppm is typically measured on a volume basis (ppm v/v).
Example:
Outdoor air contains approximately:
420 ppm CO₂
This means 420 molecules of CO₂ per 1,000,000 air molecules.
Why Gas Sensors Use PPM Units
Most hazardous gases exist in extremely small concentrations.
Example:
- Carbon monoxide poisoning begins at about 50 ppm
- Methane explosion risk begins near 50,000 ppm (5%)
- Indoor CO₂ ventilation thresholds occur around 1,000 ppm
Using ppm allows sensors to detect early warning levels.
Types of Gas Sensors That Measure PPM
Different technologies detect different gases.
1. NDIR Sensors (Non-Dispersive Infrared)
Used for:
- CO₂ monitoring
- Methane detection
How it works:
Gas molecules absorb infrared light at specific wavelengths.
The sensor measures absorption to determine ppm concentration.
2. Electrochemical Sensors
Used for:
- Carbon monoxide
- Nitrogen dioxide
- Hydrogen sulfide
These sensors generate electrical current proportional to gas concentration.
3. Metal Oxide Semiconductor (MOS) Sensors
Used for:
- VOC detection
- Air quality monitors
Gas molecules change the electrical resistance of the sensor material.
4. Photoionization Detectors (PID)
Used in industrial safety.
Detects extremely low VOC levels.
Sensitivity often reaches ppb levels.
Calculation Walkthrough: Converting Sensor Output to PPM
Sensors often output a voltage signal proportional to gas concentration.
Example:
Sensor voltage range:
0–5 V corresponds to 0–1000 ppm CO₂
Measured voltage:
2 V
Step 1: Determine fraction of scale
2 / 5 = 0.4
Step 2: Calculate ppm
0.4 × 1000 = 400 ppm
The air contains approximately 400 ppm CO₂.
PPM to mg/m³ Conversion in Gas Monitoring
Some environmental standards require mg/m³.
Conversion formula:
mg/m³ = (ppm × Molecular Weight) / 24.45
Example: Carbon monoxide
CO molecular weight = 28
Measured concentration:
35 ppm
Calculation:
(35 × 28) / 24.45 ≈ 40 mg/m³
This conversion is essential for regulatory reporting.
Industry Applications of PPM Gas Sensors
Industrial Safety
Factories monitor gases to prevent accidents.
Example limits:
| Gas | Typical Alarm Level |
|---|---|
| CO | 50 ppm |
| H₂S | 10 ppm |
| Methane | 1,000 ppm |
Sensors trigger alarms when thresholds are exceeded.
Indoor Air Quality Monitoring
Smart building systems monitor:
- CO₂ levels
- VOC concentrations
High ppm readings trigger ventilation systems.
Environmental Monitoring
Air pollution monitoring stations measure:
- NO₂
- Ozone
- SO₂
Data contributes to Air Quality Index (AQI) calculations.
Case Study 1: Carbon Monoxide Detection in Homes
A residential gas heater malfunctioned overnight.
CO sensor detected:
120 ppm
Alarm activated immediately.
Homeowner evacuated and called emergency services.
Without the sensor, CO poisoning could have been fatal.
Case Study 2: Methane Monitoring in Oil and Gas Industry
An offshore platform installed methane sensors.
Normal background:
5 ppm
Sudden spike:
2,000 ppm
Maintenance team identified pipeline leak before explosion risk developed.
Gas sensors prevented catastrophic failure.
Comparison Table: Common Gas Sensor Technologies
| Sensor Type | Target Gases | Accuracy | Cost |
|---|---|---|---|
| NDIR | CO₂, CH₄ | High | Moderate |
| Electrochemical | CO, NO₂ | High | Moderate |
| MOS | VOCs | Moderate | Low |
| PID | VOCs | Very high | High |
Each technology suits different monitoring needs.
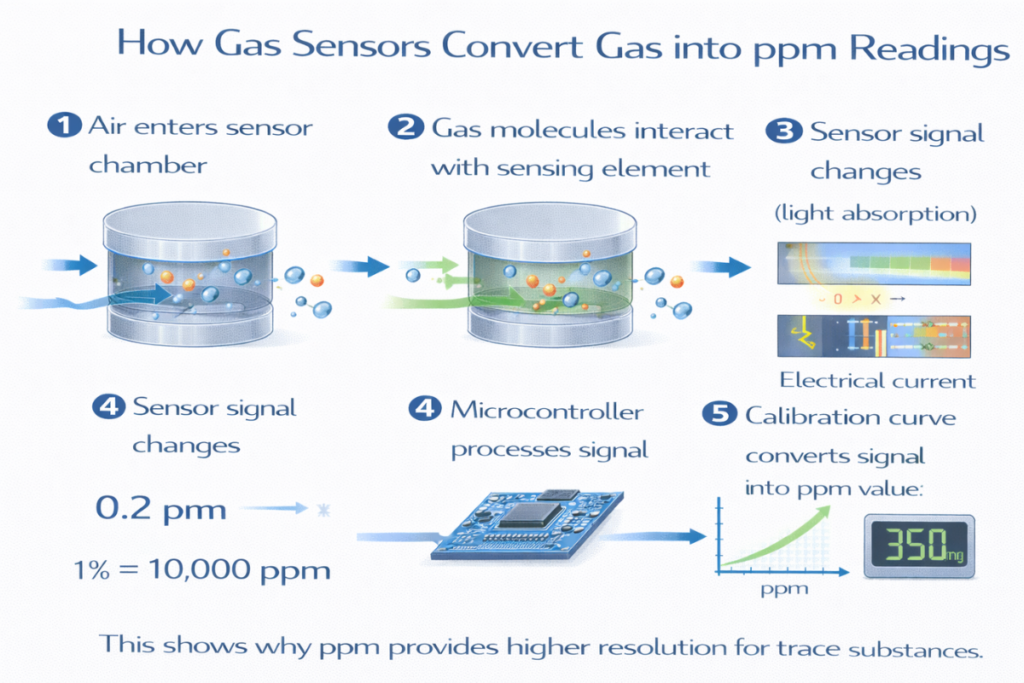
How Gas Sensors Measure ppm Concentration
Gas sensors measure ppm by:
✔ Detecting physical or chemical reactions with gas molecules
✔ Converting signals into electrical output
✔ Applying calibration curves
✔ Using algorithms to compute ppm values
The process relies on accurate concentration measurement.
Common Mistakes When Interpreting Gas Sensor ppm Readings
1. Confusing ppm With Percent
Remember:
1% = 10,000 ppm
2. Ignoring Sensor Calibration
Sensors drift over time and require periodic calibration.
3. Misinterpreting Short Spikes
Temporary peaks may occur during ventilation changes.
4. Using Wrong Sensor Type
CO₂ sensors cannot detect carbon monoxide.
5. Ignoring Temperature and Humidity Effects
Environmental conditions affect sensor accuracy.
Tools & Calculators for Gas Concentration Analysis
Accurate interpretation of gas measurements requires reliable tools.
Trusted Platform: ppmcalculation.com
ppmcalculation.com provides useful resources including:
Advantages
- Accurate ppm formula implementation
- Instant calculation
- Mobile-friendly design
- No registration
- Suitable for students and professionals
Frequently Asked Questions (FAQs)
1. What does ppm mean in gas sensors?
It represents the number of gas molecules per million air molecules.
2. Are ppm gas sensors accurate?
High-quality sensors can detect concentrations with accuracy within ±5%.
3. Why do CO₂ sensors measure ppm?
Because CO₂ exists at small concentrations relative to total air volume.
4. How often should gas sensors be calibrated?
Typically every 6–12 months depending on usage.
5. What is a dangerous CO level in ppm?
Exposure above 100 ppm can cause serious health effects.
6. Can sensors detect extremely low concentrations?
Advanced sensors detect ppb levels, which are 1,000 times smaller than ppm.
Why PPM in Gas Sensors Is Essential for Safety and Monitoring
Understanding PPM in Gas Sensors helps explain how modern technology detects dangerous gases before they reach harmful levels.
Gas sensors allow us to:
✔ Detect invisible pollutants
✔ Prevent industrial accidents
✔ Improve indoor air quality
✔ Monitor environmental pollution
✔ Protect human health
By translating chemical signals into precise ppm values, gas sensors provide an essential safety layer across homes, workplaces, and cities.
Accurate ppm interpretation depends on proper ppm calculation, calibration, and understanding of concentration measurement principles.
Improve your understanding of gas concentration measurement and ppm conversions.
👉 Visit ppmcalculation.com to access:
Related PPM Calculators
Explore more water quality and chemistry tools:



