Why ppm in Aquarium Water Quality Is Critical for Fish Health
Aquarium fish may look peaceful, but their survival depends on precise water chemistry. Invisible toxins such as ammonia and nitrite can rise silently, measured only in parts per million (ppm)—yet even small increases can become deadly.
Understanding how ppm measurements help with aquarium water quality is essential for both beginner hobbyists and advanced aquarists. A difference between 0.25 ppm and 1 ppm ammonia may mean the difference between a thriving tank and sudden fish loss.
Aquarium water testing relies heavily on accurate ppm calculation, proper concentration measurement, and correct interpretation of values like:
- Ammonia (NH₃/NH₄⁺)
- Nitrite (NO₂⁻)
- Nitrate (NO₃⁻)
- pH
- Total Dissolved Solids (TDS)
In this detailed guide, we will explain how ppm works in aquariums, what safe ranges are, how to calculate dosing and water changes, and how to avoid common mistakes.
What Does ppm Mean in Aquarium Water Testing?
Definition of ppm in Aquariums
In aquarium systems, ppm represents milligrams of a dissolved substance per liter of water.
For freshwater tanks:
1 ppm ≈ 1 mg/L
Because 1 liter of water weighs approximately 1 kilogram, ppm and mg/L are practically interchangeable.
This relationship simplifies ppm calculation in aquarium care.
Why ppm Matters in Fish Tanks
Fish constantly release waste. Uneaten food decomposes. Bacteria convert toxins through the nitrogen cycle. Each step is measured in ppm.
Even tiny concentrations affect:
✔ Fish respiration
✔ Gill health
✔ Immune function
✔ Stress levels
✔ Plant growth
Aquarium ecosystems are closed systems—small changes amplify quickly.
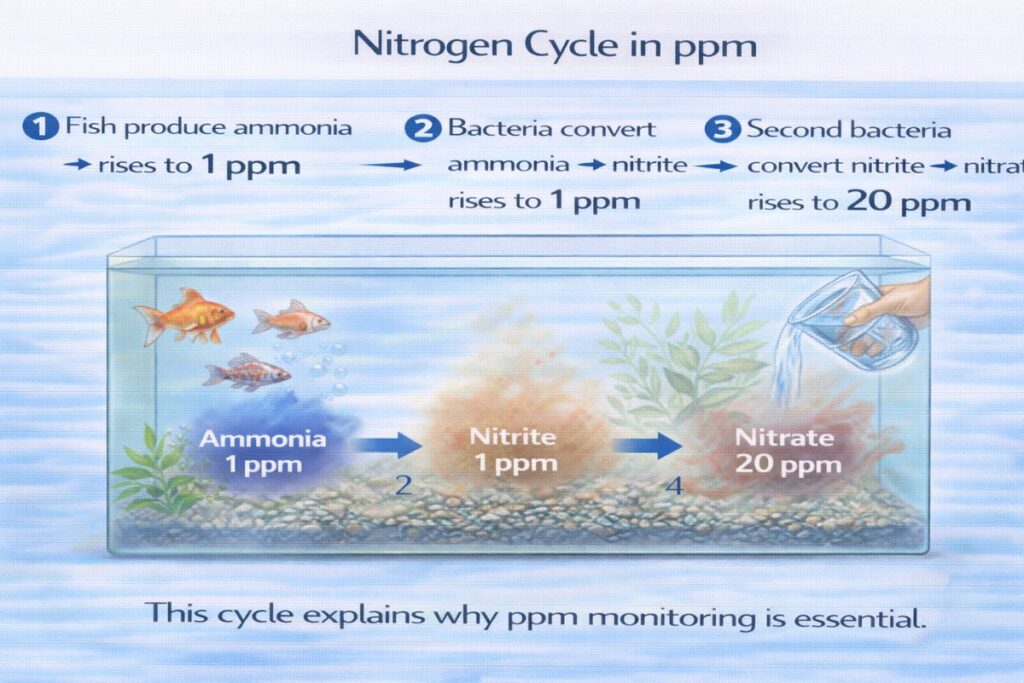
The Nitrogen Cycle: The Foundation of Aquarium ppm Management
Step 1: Ammonia Formation
Fish waste produces ammonia.
Safe level:
0 ppm (ideal)
Danger begins above 0.25 ppm.
Step 2: Nitrite Conversion
Beneficial bacteria convert ammonia into nitrite.
Safe level:
0 ppm (ideal)
Nitrite interferes with oxygen transport in fish blood.
Step 3: Nitrate Formation
Another bacterial group converts nitrite into nitrate.
Safe range:
0–40 ppm (freshwater)
Marine tanks prefer:
<20 ppm
Safe ppm Ranges for Aquarium Parameters
| Parameter | Ideal ppm | Danger Zone |
|---|---|---|
| Ammonia | 0 ppm | >0.25 ppm |
| Nitrite | 0 ppm | >0.5 ppm |
| Nitrate | <40 ppm (freshwater) | >80 ppm |
| TDS | 100–400 ppm (freshwater) | >600 ppm |
Calculation Walkthrough: Water Change to Reduce Nitrate
Scenario
Tank size = 100 liters
Current nitrate = 80 ppm
Target nitrate = 20 ppm
Step 1: Determine Reduction Needed
80 ppm → 20 ppm
Reduction = 60 ppm
Step 2: Calculate Percentage Water Change
Required reduction fraction:
60 / 80 = 0.75
This means 75% water change required.
Practical Advice
Instead of a single 75% change (stressful), perform:
- Two 40% water changes over 48 hours
ppm Calculation Example: Dosing Dechlorinator
Suppose:
Chlorine concentration = 2 ppm
Tank volume = 50 liters
If product neutralizes 1 ppm per 1 mL per 40 liters:
Step 1: Determine chlorine load
2 ppm × 50 L = 100 mg chlorine
Step 2: Dose proportionally
Required = 2.5 mL
Using a ppm converter simplifies dosing.
Case Study 1: Ammonia Spike After Adding New Fish
A beginner added five new fish to a 60 L tank.
After 48 hours:
Ammonia = 1 ppm
Symptoms:
- Fish gasping
- Red gills
- Lethargy
Cause:
Tank not fully cycled.
Solution:
- Immediate 50% water change
- Added beneficial bacteria
- Reduced feeding
Ammonia returned to 0 ppm in 5 days.
Case Study 2: Nitrate Control in Planted Tank
A planted 200 L aquarium showed:
Nitrate = 90 ppm
Plants thriving but fish stressed.
Action:
- Increased weekly water change from 20% to 40%
- Added fast-growing stem plants
Result:
Nitrate stabilized at 25 ppm.
Aquarium Industry Example: Commercial Fish Farms
Commercial aquaculture monitors:
- Ammonia at <0.02 ppm (unionized NH₃)
- Nitrite at <0.1 ppm
Even small ppm changes affect survival rates and economic yield.
Precision ppm monitoring is critical in fish farming.
Freshwater vs Saltwater ppm Management
| Parameter | Freshwater | Saltwater |
|---|---|---|
| Ammonia | 0 ppm | 0 ppm |
| Nitrite | 0 ppm | 0 ppm |
| Nitrate | <40 ppm | <20 ppm |
| TDS | 100–400 ppm | 35,000 ppm (salt) |
Saltwater tanks have much higher total dissolved solids but similar nitrogen limits.

How to Test ppm in Aquarium Water
1. Liquid Test Kits
✔ Most accurate for hobbyists
✔ Measures ammonia, nitrite, nitrate
2. Test Strips
✔ Fast
✘ Slightly less accurate
3. Digital Meters
✔ Advanced users
✔ Direct concentration measurement
How to Lower Ammonia ppm in Aquarium Fast
✔ Immediate 50% water change
✔ Stop feeding for 24–48 hours
✔ Add nitrifying bacteria
✔ Improve filtration
✔ Increase aeration
Quick action prevents fish loss.
Common Mistakes in Aquarium ppm Management
1. Overfeeding
Increases ammonia quickly.
2. Overcleaning Filter Media
Kills beneficial bacteria.
3. Ignoring Gradual Nitrate Buildup
Nitrate creeps up slowly over weeks.
4. Misreading Test Colors
Lighting affects visual interpretation.
5. Confusing ppm With Percentage
1% ammonia = 10,000 ppm (lethal).
How ppmcalculation.com Helps Aquarium Owners
Although primarily known for industrial and chemical concentration tools, ppmcalculation.com provides valuable resources for:
✔ ppm calculation
✔ ppm to mg/L conversion
✔ ppm converter tools
✔ dilution estimation
✔ concentration measurement guidance
These tools simplify dosing calculations and water change planning.
FAQs: Aquarium ppm Questions Answered
1. What is a safe ammonia ppm in aquariums?
0 ppm is ideal.
2. How often should I test ppm?
Weekly for stable tanks; daily during cycling.
3. Can nitrate ever be zero?
Possible in heavily planted or lightly stocked tanks.
4. Does higher TDS always mean bad water?
Not necessarily; context matters.
5. Why is nitrite dangerous?
It reduces oxygen transport in fish blood.
6. Can ppm fluctuate daily?
Yes, especially in newly established tanks.
Why ppm Measurements Are the Key to Aquarium Water Quality
Understanding how ppm measurements help with aquarium water quality empowers fish keepers to maintain healthy aquatic ecosystems.
Key Takeaways
✔ Ammonia and nitrite must remain at 0 ppm
✔ Nitrate should stay below 40 ppm (freshwater)
✔ ppm calculation guides water changes
✔ Accurate concentration measurement prevents fish stress
✔ Digital tools improve precision
Small ppm differences have big biological consequences in closed aquatic systems.
Related PPM Calculators
Explore more water quality and chemistry tools:



